 In this trial, a total of 1,444 vaccine recipients were followed for safety for at least 2 months after the second dose.Ĭommonly reported side effects in the clinical trial included injection site pain (sore arm), redness and swelling, fatigue, headache, muscle and/or joint pain, chills, fever, swollen lymph nodes, nausea and decreased appetite. The available safety data to support the EUA include more than 4,600 participants (3,100 vaccine, 1,538 placebo) ages 5 through 11 years enrolled in the ongoing study. In this analysis, among participants without evidence of prior infection with SARS-CoV-2, 3 cases of COVID-19 occurred among 1,305 vaccine recipients and 16 cases of COVID-19 occurred among 663 placebo recipients the vaccine was 90.7% effective in preventing COVID-19. The FDA also conducted a preliminary analysis of cases of COVID-19 occurring seven days after the second dose. 
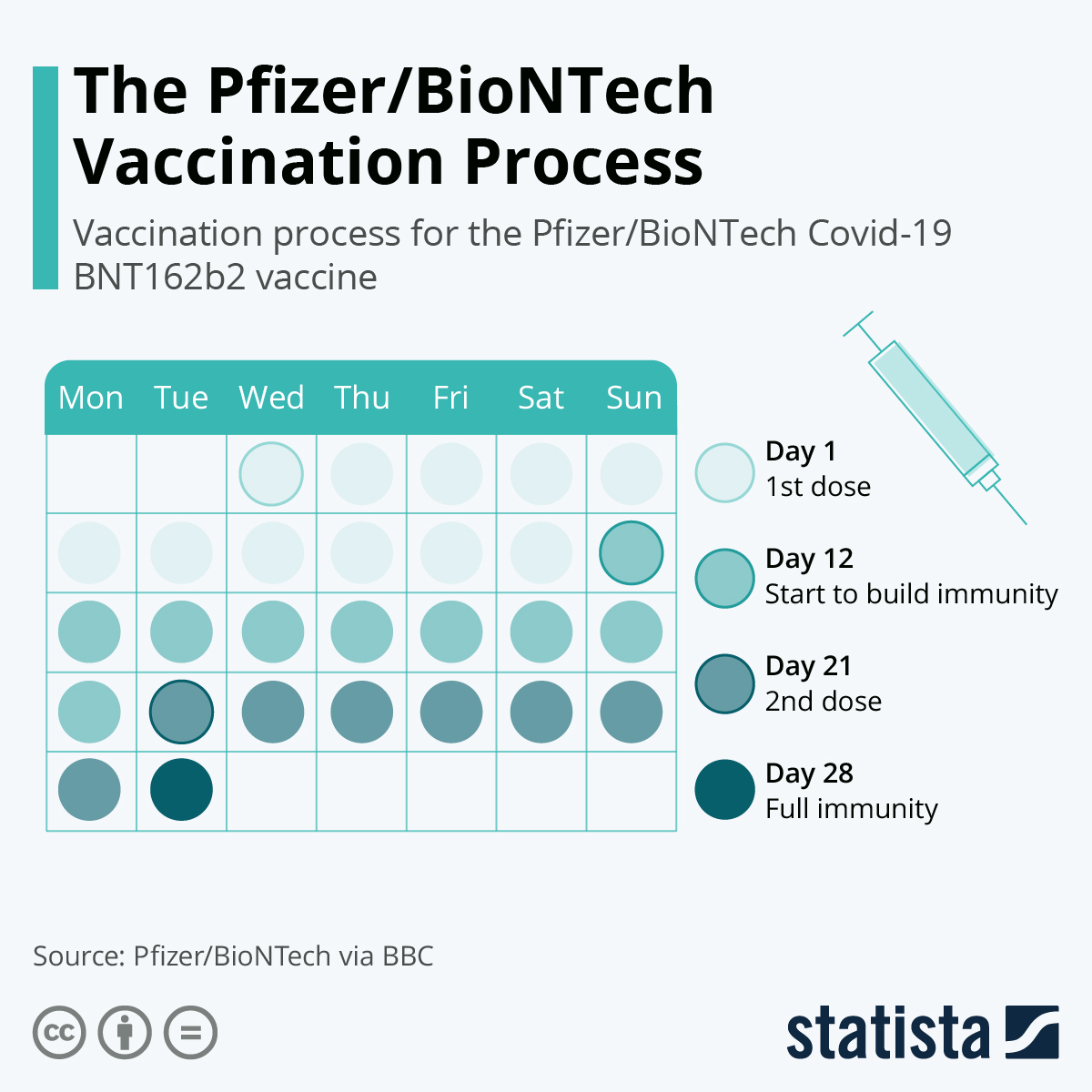
The immune responses of the younger age participants were comparable to the older participants. The FDA analyzed data that compared the immune response of 264 participants from this study to 253 participants 16 through 25 years of age who had two higher doses of the vaccine in a previous study which determined the vaccine to be effective in preventing COVID-19. Children in the vaccine group received two doses of the Pfizer-BioNTech COVID-19 Vaccine containing 10 micrograms of messenger RNA per dose. The study is being conducted in the U.S., Finland, Poland and Spain. The effectiveness data to support the EUA in children down to 5 years of age is based on an ongoing randomized, placebo-controlled study that has enrolled approximately 4,700 children 5 through 11 years of age. Based on the totality of scientific evidence available, the known and potential benefits of the Pfizer-BioNTech COVID-19 vaccine in individuals down to 5 years of age outweigh the known and potential risks.įDA Evaluation of Available Effectiveness Data The FDA has determined this Pfizer vaccine has met the criteria for emergency use authorization. We hope this information helps build confidence of parents who are deciding whether to have their children vaccinated,” said Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research. As part of our commitment to transparency around our decision-making, which included our public advisory committee meeting earlier this week, we have posted documents today supporting our decision and additional information detailing our evaluation of the data will be posted soon. We are confident in the safety, effectiveness and manufacturing data behind this authorization. “The FDA is committed to making decisions that are guided by science that the public and healthcare community can trust. in individuals less than 18 years of age, with 146 deaths in the 5 through 11 years age group. 17, 691 deaths from COVID-19 have been reported in the U.S. According to the CDC, approximately 8,300 COVID-19 cases in children 5 through 11 years of age resulted in hospitalization. In the U.S., COVID-19 cases in children 5 through 11 years of age make up 39% of cases in individuals younger than 18 years of age. The Pfizer-BioNTech COVID-19 Vaccine for children 5 through 11 years of age is administered as a two-dose primary series, 3 weeks apart, but is a lower dose (10 micrograms) than that used for individuals 12 years of age and older (30 micrograms). “Our comprehensive and rigorous evaluation of the data pertaining to the vaccine’s safety and effectiveness should help assure parents and guardians that this vaccine meets our high standards.” Vaccinating younger children against COVID-19 will bring us closer to returning to a sense of normalcy,” said Acting FDA Commissioner Janet Woodcock, M.D. “As a mother and a physician, I know that parents, caregivers, school staff, and children have been waiting for today’s authorization.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
